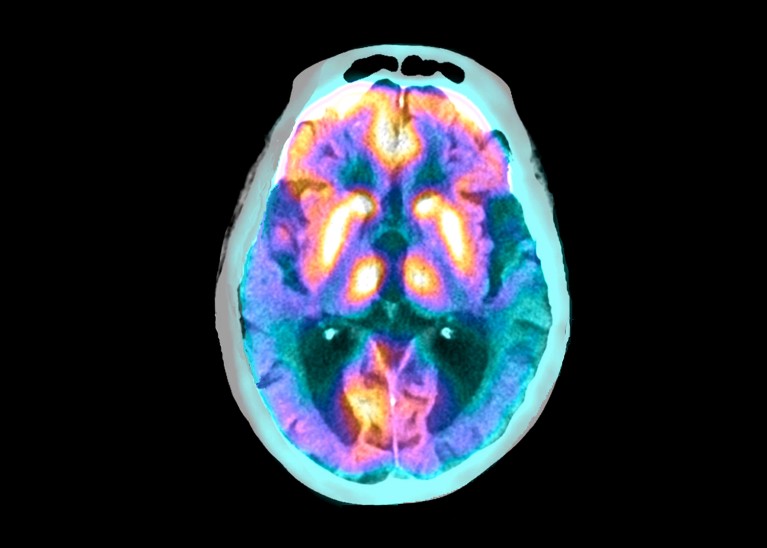
Superimposed scans show the brain of a person with Alzheimer’s disease. Credit: Zephyr/Science Photo Library
A drug for Alzheimer’s disease has won unanimous approval from independent scientists advising the US Food and Drug Administration (FDA), bringing the treatment closer to use in the clinic.
The drug, donanemab, would be the second on the US market that slows the cognitive decline caused by the disease. But donanemab’s effects are modest, it does not reverse symptoms and the FDA might limit who can take it.
At a 10 June meeting at the FDA’s headquarters in Silver Spring, Maryland, all 11 members of an FDA independent scientific advisory committee voted that donanemab, made by Eli Lilly, based in Indianapolis, Indiana, is effective at treating Alzheimer’s, at least in people at early stages of the disease, and that its benefits outweigh its risks.
The advisory meeting itself was a surprise to many observers, who had expected the FDA to quickly approve donanemab without convening an advisory committee. Instead the FDA delayed its decision until after a public meeting could be held because of questions about the drug’s efficacy in people with certain markers of Alzheimer’s disease. But in the end, “it was a very positive meeting,” says neurologist David Knopman of the Mayo Clinic in Rochester, Minnesota, who was not on the committee. “It would have been difficult for someone to object based on the standard of the data.”
Amyloid attacker
Donanemab is an antibody that attacks amyloid, a sticky protein that accumulates in the brains of people with Alzheimer’s. In data submitted to the FDA, Lilly reported that the 860 trial participants who received donanemab lost their cognitive abilities more slowly over the course of 18 months than those who received a placebo. The drug, however, did not reverse the disease. Research1,2 shows that donanemab slows symptoms roughly as well as rival drug lecanemab, which also attacks amyloid. It is made by bio-pharmaceutical companies Eisai in Tokyo and Biogen in Cambridge, Massachusetts.
More Alzheimer’s drugs head for FDA review: what scientists are watching
Unlike previous trials of monoclonal antibodies, Lilly only tested people whose brains contained both amyloid and another protein called tau that is associated with cognitive decline. Donanemab seemed more effective in people who had low to moderate tau levels at the start of the trial than in people who had high levels. But the FDA pointed out that the disease might have progressed more slowly in the lower-tau group than it in those with higher tau.
At the meeting, advisory committee members were broadly supportive of the drug. Some noted, however, that Lilly has little evidence that it works in people with no to very little tau. But the committee decided against restricting the drug’s usage on the basis of tau levels, because screening for tau is complex and costly. A screening requirement would prevent an unacceptably high number of people from accessing the drug, the committee decided.
The panel members also expressed some concerns about a condition called amyloid-related imaging abnormalities (ARIA) that can cause brain bleeding and seizures. ARIA, which can be fatal, is thought to occur when the antibodies weaken blood vessels in the brain. Lilly recorded more ARIA-related deaths among people who received the drug than in the placebo group. Lecanemab also has been linked to ARIA, but the increased risk seems to be much higher with donanemab3.
Controversial therapies
The approval is a bright spot for amyloid-targeting Alzheimer’s drugs after numerous controversies. The FDA approved the first such drug, Biogen and Eisai’s aducanumab, in 2021 over the objections of its advisory committee, leading three committee members to resign in protest. A US Congressional investigation found that that the FDA had improperly guided the manufacturers through the approval process. Many insurers were unconvinced by the drug’s efficacy, and most refused to cover it. Biogen stopped making the drug this year. And three people died from ARIA-related conditions during clinical trials of lecanumab.
Landmark Alzheimer’s drug approval confounds research community
The donanemab committee said that more research is still needed on the drug, including how long people should take it, and its efficacy in people with different levels of tau. Knopman says that it remains to be seen whether the drug’s modest effect will persist for years.
The committee also recommended more research on the drug’s efficacy in people of colour — more than 90% of Lilly’s trial participants were white — and people with Down’s syndrome or genetic mutations that make them more prone to ARIA. Committee member Kathleen Poston, a neurologist at Stanford University Medical Center in California, says scientists need to obtain these data “to make sure that these encouraging findings can be extrapolated to everyone with Alzheimer’s disease.” The FDA’s final decision is expected later this year.